Questions
- What is the Valence of an Atom?
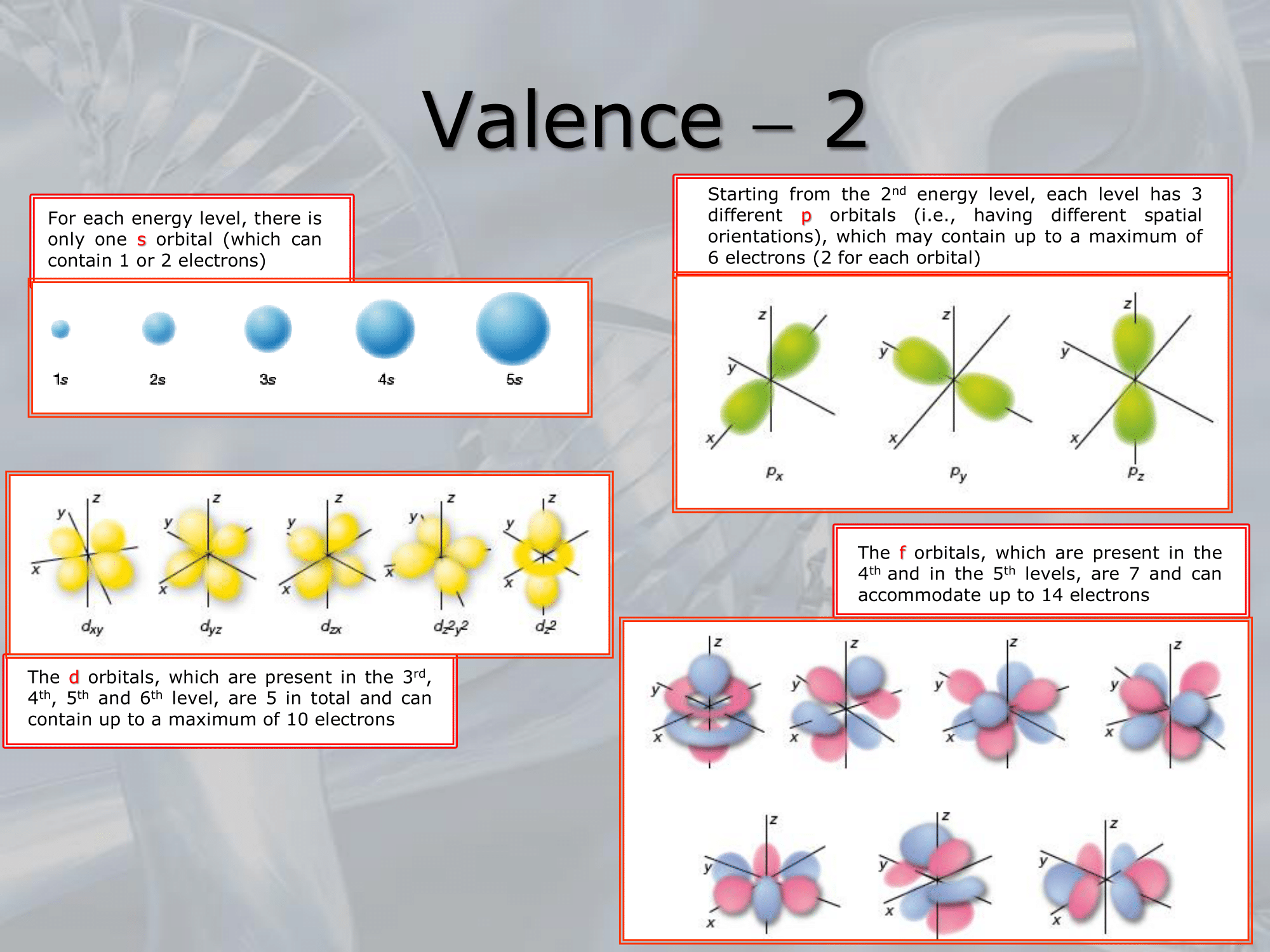
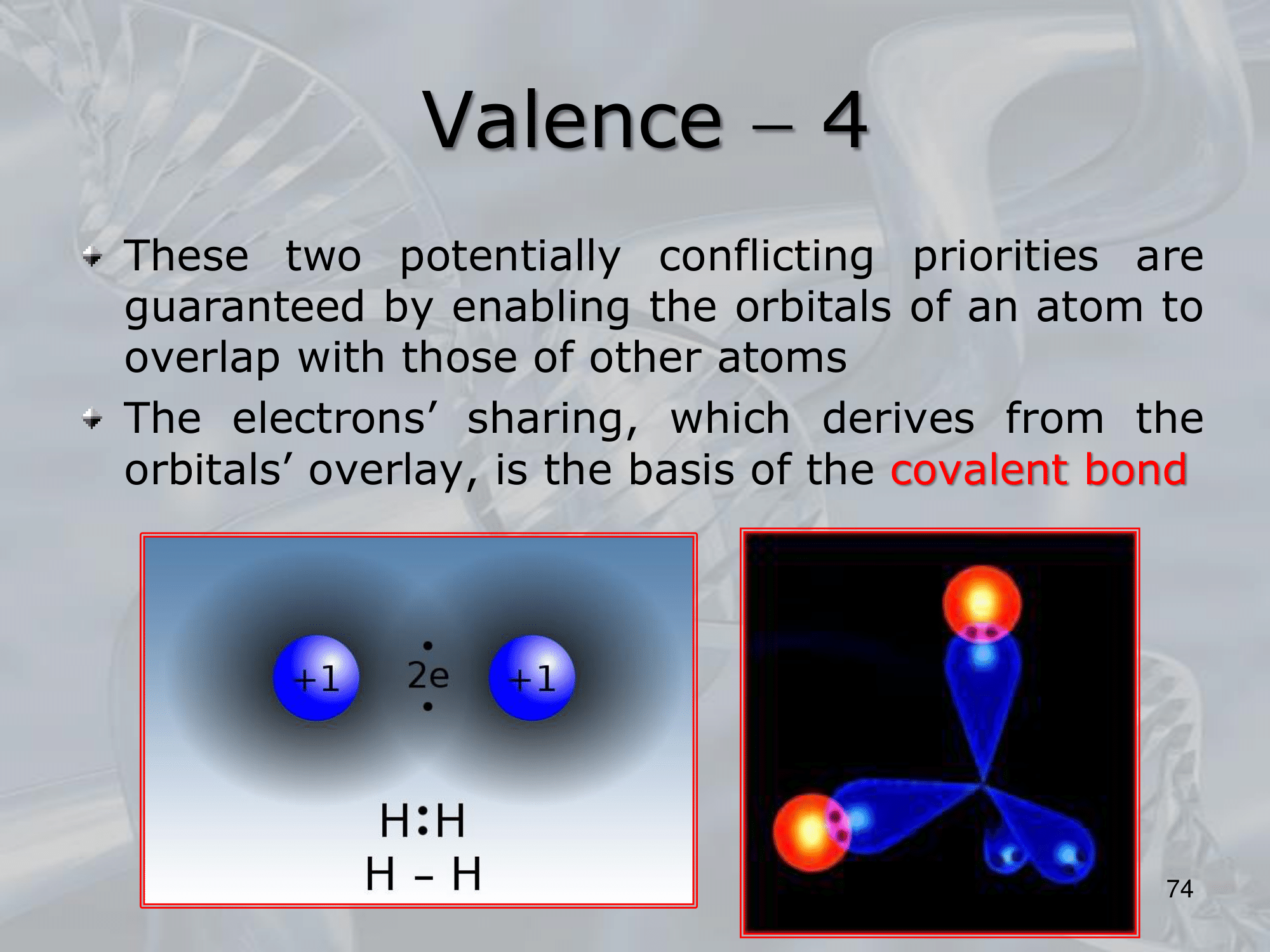
- ==Valence refers to the number of electrons that an atom can share, donate or accept to form chemical bonds with other atoms.
In other words, it is the number of unpaired electrons in the outermost energy level (also called valence shell) of an atom==.
The valence electrons are involved in chemical reactions, and the number of valence electrons determines an atom’s chemical properties, such as its reactivity, electronegativity, and ability to form bonds with other atoms. - For example, carbon has four valence electrons in its outermost energy level, which allows it to form covalent bonds with up to four other atoms.
Similarly, oxygen has six valence electrons and can form two covalent bonds with other atoms to complete its octet. - The concept of valence is fundamental to understanding chemical bonding and the behavior of atoms in chemical reactions.
By knowing the number of valence electrons in different elements, chemists can predict how they will interact with each other and form stable compounds.
- ==Valence refers to the number of electrons that an atom can share, donate or accept to form chemical bonds with other atoms.
IMPORTANTE
Slides with Notes