Intermolecular Forces
We have seen three types of intermolecular forces:
- Dipole-Dipole Interaction (between polar molecules)
- Hydrogen Bonding (the strongest, between polar molecules with OH, NH, or FH groups)
- London Dispersion Forces (the weakest, between non-polar molecules)
- Ion-Dipole Interaction, which can be differentiated in :
- Ion Dipole Forces (for example between water and salt, where water is a polar molecule)
- Ion Induced Dipole Force (between an ion and a non-polar molecule, in this case the ion induce a dipole and then bonds)
- Ion-Ion Interaction.
Dipole-Dipole Interaction
Usually occurs between polar molecules, let’s take for example the acetone molecule ( CHO )
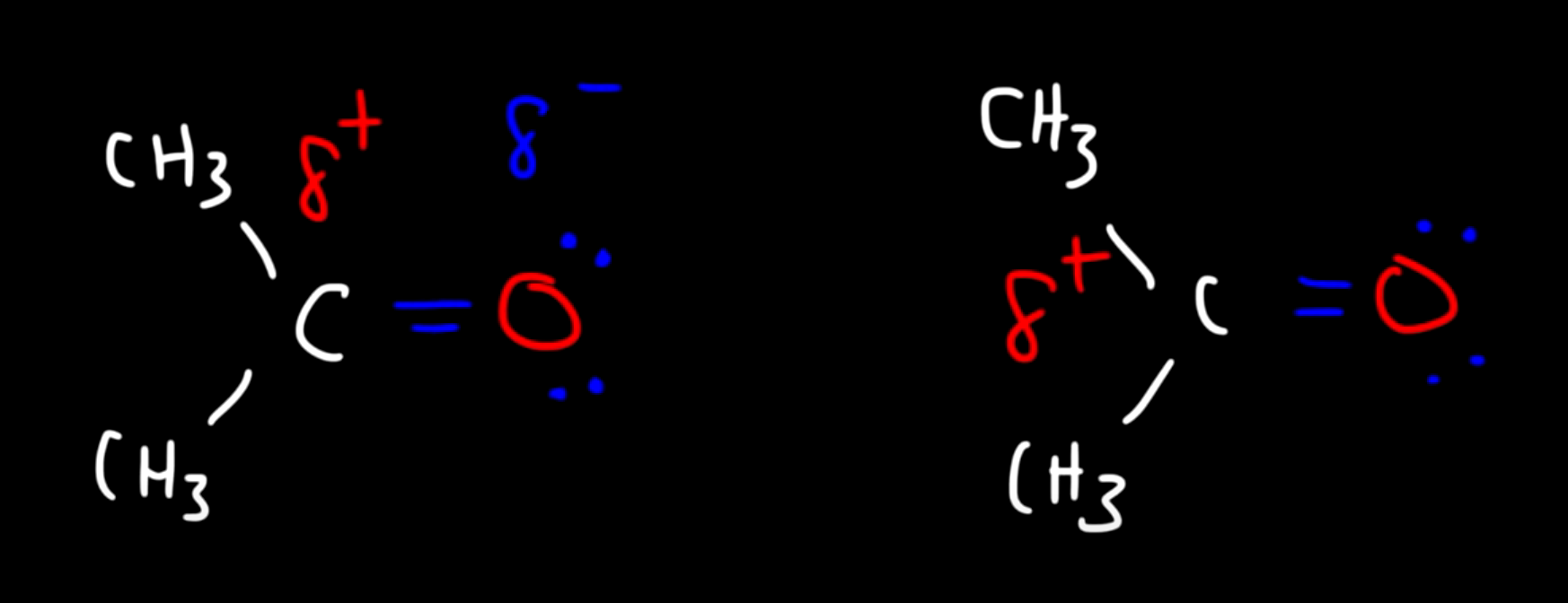 The Interaction between these two molecules is defined as dipole-dipole (the positive pole of one molecule is attracted to the negative pole of the other)
The Interaction between these two molecules is defined as dipole-dipole (the positive pole of one molecule is attracted to the negative pole of the other)
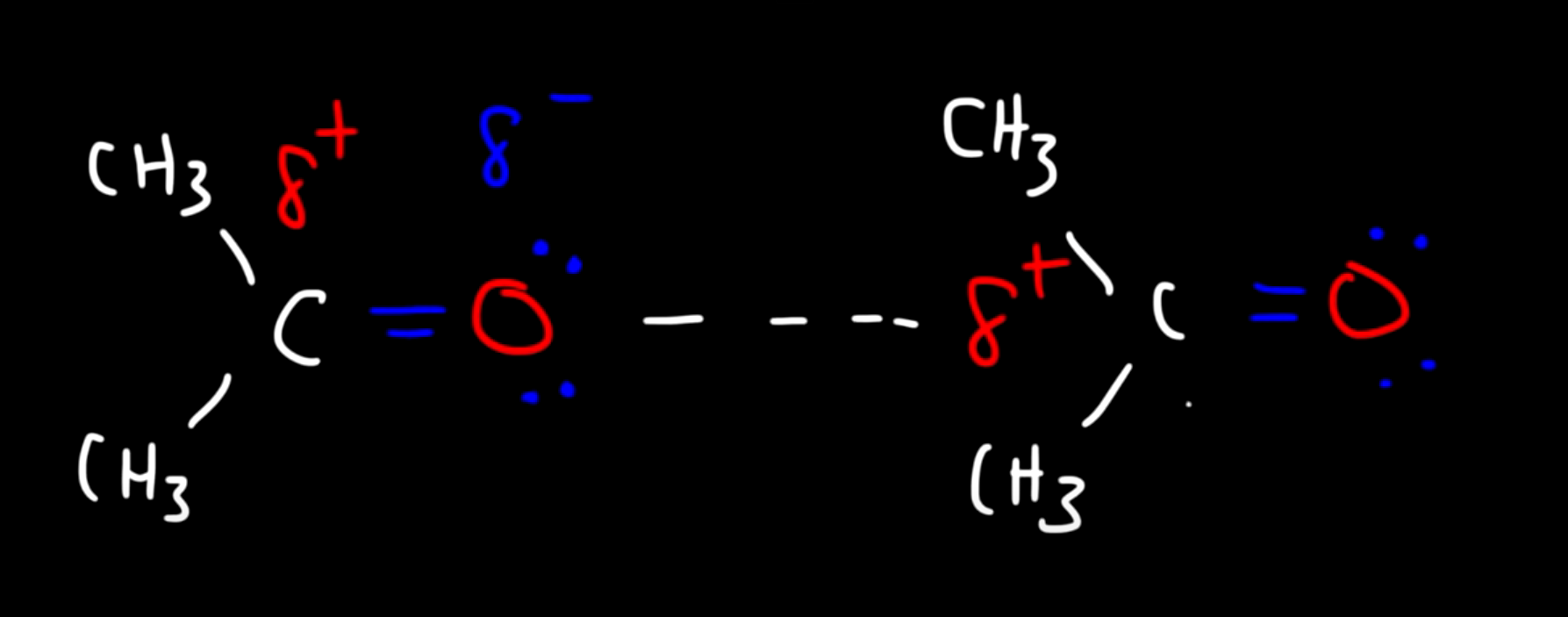
Another example:
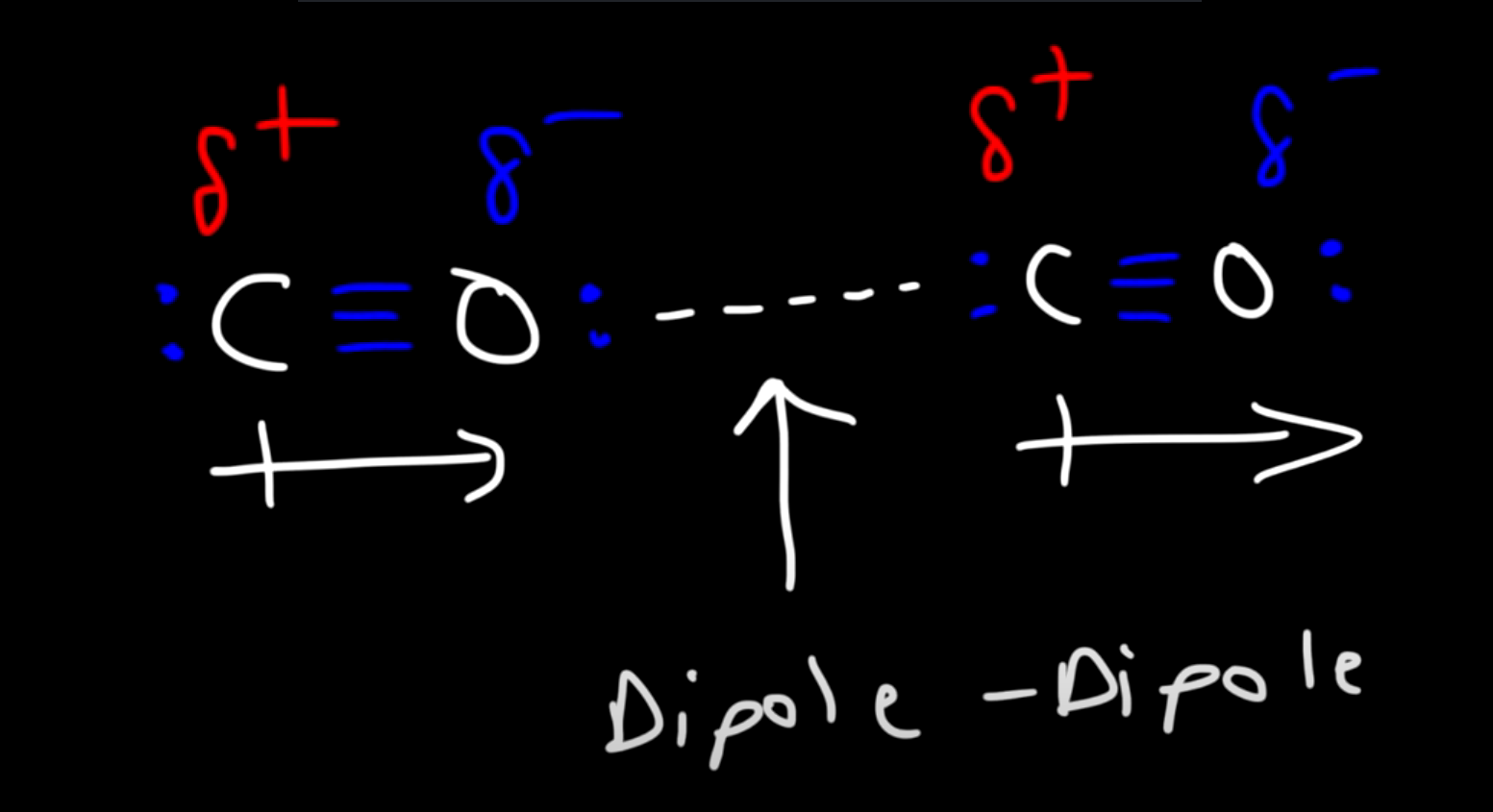
Hydrogen Bonding
A special type of dipole-dipole interaction, it occurs when hydrogen bonds either with Oxygen (O), Nitrogen (N) or Fluorine (F).
Let’s take for example the water molecule ( HO )
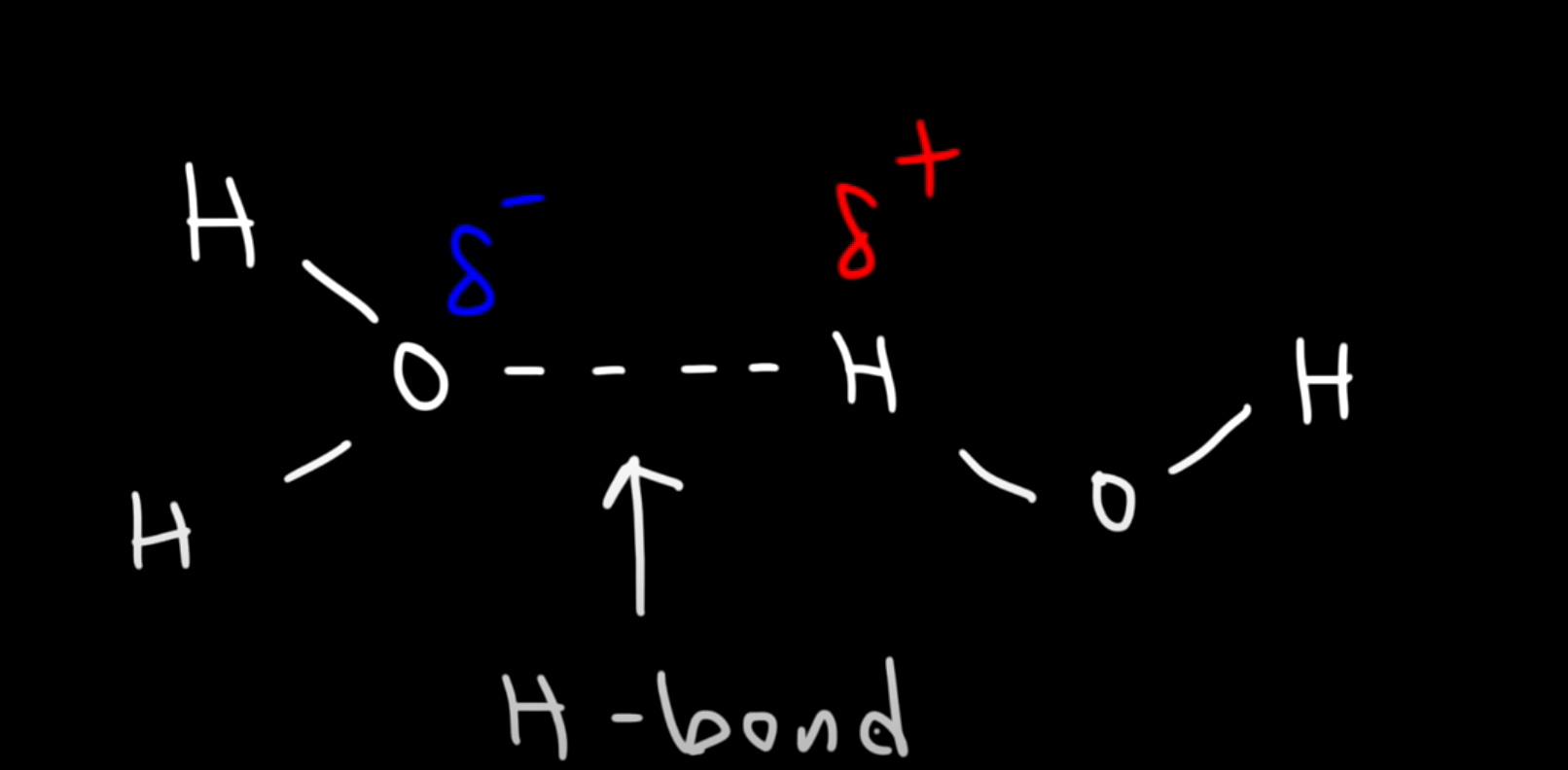
Molecules with hydrogen bonds have:
- higher boiling points
- higher solubility (for example methanol ( CHOH ) mixes completely with water)
With respect to molecules that do not have hydrogen bonds.
Solubility and Boiling Points
- Molecules with hydrogen bonds have both higher boiling points and higher solubility with respect to non-hydrogen bonds molecule.
- Between two different hydrogen bond molecule the larger one will have higher boiling point, and the more polar one will have higher solubility.
~Ex.: Ethanol ( CHCHOH ) vs. Dimethyl Ether ( CHOCH ) Ethanol has the OH group, so it will have an hydrogen bond, while Dimethyl Ether will not, so the Ethanol has both higher boiling points and higher solubility.
~Ex.: Ethanol ( CHCHOH ) vs. Butanol ( CHCHCHCHOH ) They have both the OH group so they both have the hydrogen bond, but butanol has a much bigger hydrocarbon chain, it has more atoms in each molecules, as a result it will have more London Dispersion Forces (in addition to the hydrogen bond), so it will have higher boiling point. While the OH group is polar, the CH group is non-polar, this is why the ethanol is more soluble than butanol (it is more polar overall).
London Dispersion Forces
We can find these forces in every atom and molecule, however they are the predominant forces of non-polar molecules.
Starting from a non polar molecule (let’s take an atom for example, the same applies to a molecule), we start with a non-polar situation.
 (The “+6” indicates that there are 6 proton in the nucleus, so there is a balance of charges)
(The “+6” indicates that there are 6 proton in the nucleus, so there is a balance of charges)
Now due to the randomness of the electrons there could be a moment where this atom in polar, when we have more electrons in the left part of the atom for example, we define it as polarized:
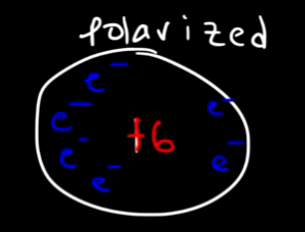
This forms a dipole that attracts other atoms:
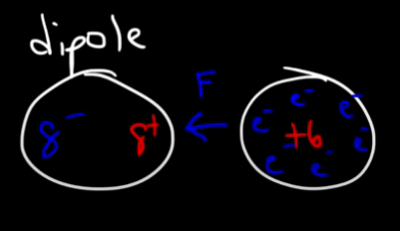
And since the electrons in the second atom are shifted, the second atom becomes polarized as well and we call this and “induced dipole”:
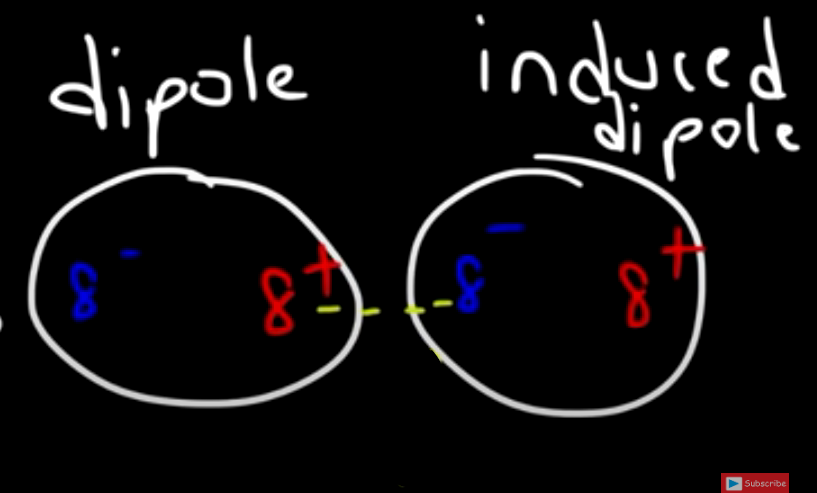
The forces that is then created (the yellow dotted line) is called the London Dispersion Force.