Questions
- What are the Tetriary and Quaternary Strucure of Proteins?
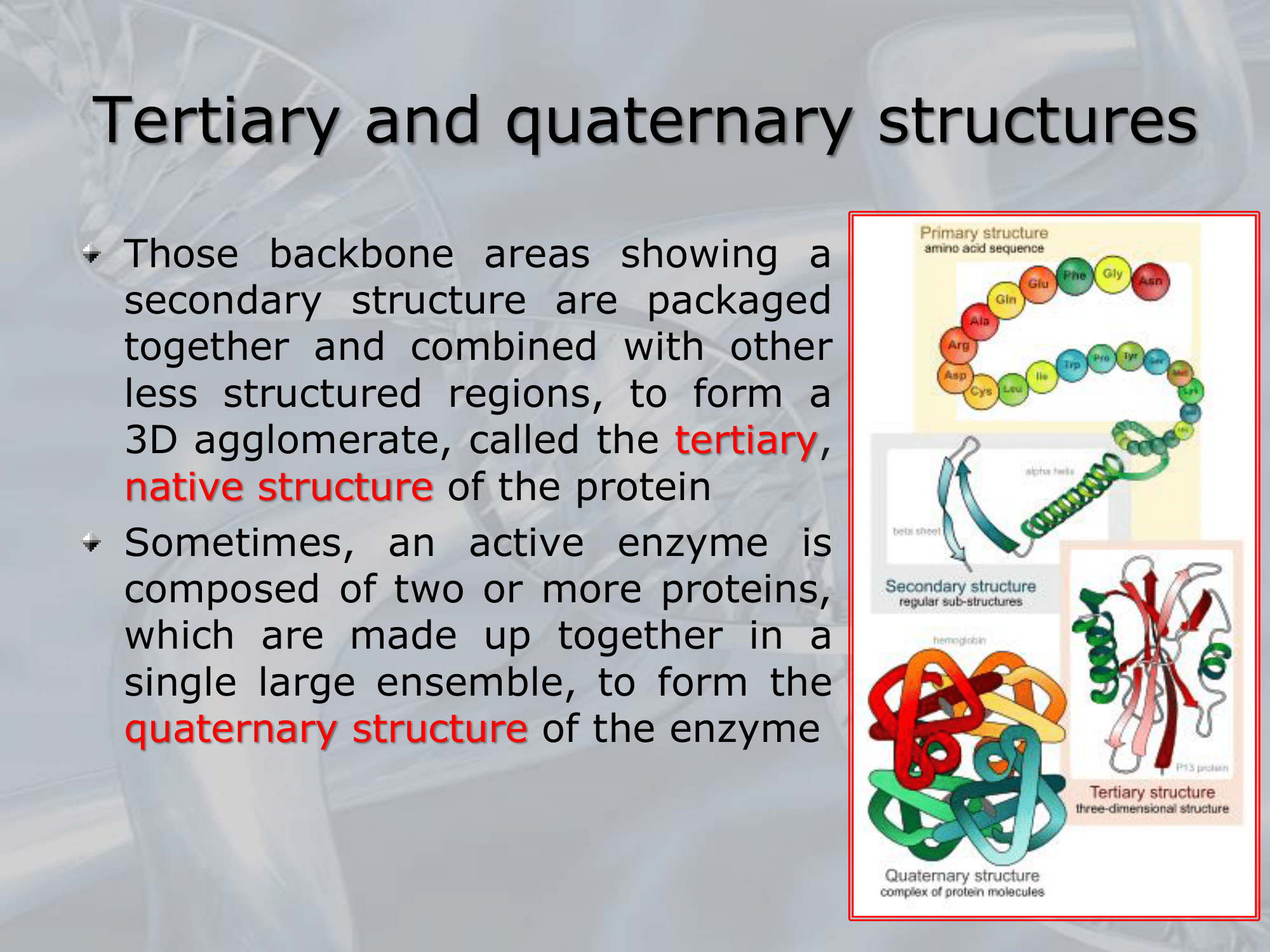
- Proteins can have up to four levels of structural organization: primary, secondary, tertiary, and quaternary.
- ==The tertiary structure of a protein refers to its three-dimensional conformation, or the way in which its secondary structural elements (alpha helices, beta sheets, and loops) are folded and arranged in space.
Tertiary structure is determined by a combination of covalent bonds (such as disulfide bonds), electrostatic interactions, hydrogen bonding, and hydrophobic interactions between amino acid side chains.
The tertiary structure is critical for a protein’s function, as it determines its binding sites and catalytic activity==. - ==The quaternary structure of a protein refers to the way in which multiple protein subunits come together to form a larger functional complex==.
Some proteins, such as hemoglobin and collagen, are made up of multiple subunits that interact with each other through noncovalent interactions, such as hydrogen bonding and electrostatic interactions.
The quaternary structure is also important for protein function, as it can affect the stability, specificity, and activity of the protein complex. - In summary, the tertiary structure of a protein refers to its three-dimensional conformation, while the quaternary structure refers to the way in which multiple protein subunits come together to form a larger functional complex.
Both levels of organization are critical for a protein’s function, and are determined by a combination of covalent and noncovalent interactions between amino acid residues.
- What are Chaperones?
- Chaperones are a class of proteins that help other proteins to fold correctly into their functional three-dimensional structures.
They play a critical role in preventing misfolding and aggregation of proteins, which can lead to diseases such as Alzheimer’s and Huntington’s. - Chaperones work by binding to partially folded or unfolded proteins and stabilizing them in a conformation that allows them to continue folding correctly.
They can also help to target misfolded or aggregated proteins for degradation by the cell's quality control machinery. - There are several families of chaperones, including the Hsp70/Hsp40 system, the chaperonin family (which includes GroEL and GroES in bacteria and CCT/TRiC in eukaryotes), and the small heat shock proteins (sHsps).
Each family has its own specific mechanisms and targets for assisting in protein folding. - Chaperones are essential for the proper functioning of cells, and their dysfunction has been implicated in a variety of diseases, including neurodegenerative disorders, cancer, and cardiovascular diseases.
As such, they have become important targets for drug development, and several chaperone-targeting drugs are currently in clinical trials for various diseases.
- Chaperones are a class of proteins that help other proteins to fold correctly into their functional three-dimensional structures.
—————————————————————
IMPORTANTE
IMPORTANTE Forces guiding protein folding:
- Electrostatic forces
- Hydrogen bonds
- Van der Waals forces
- Covalent bonds between cysteines
IMPORTANTE The Van der Waals forces are weak intermolecular attraction caused by induced molecular dipoles.
—————————————————————
Slides with Notes



IMPORTANTE Forces guiding protein folding:
- Electrostatic forces
- Hydrogen bonds
- Van der Waals forces
- Covalent bonds between cysteines
IMPORTANTE The Van der Waals forces are weak intermolecular attraction caused by induced molecular dipoles.