Questions
- What is an Hydrogen Bond?
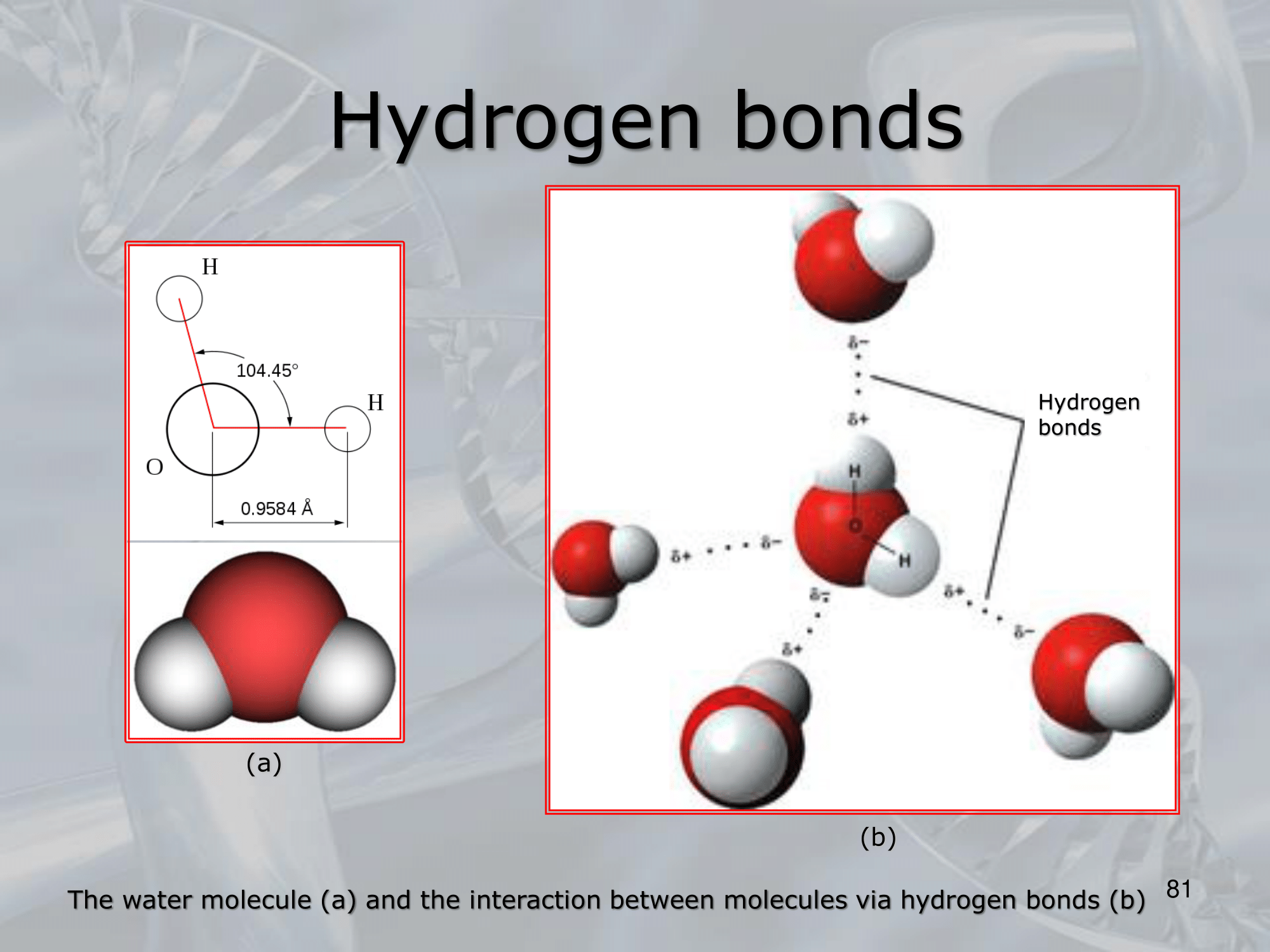
- An hydrogen bond is a type of non-covalent chemical bond that occurs between a hydrogen atom that is covalently bonded to a highly electronegative atom, such as oxygen or nitrogen, and another electronegative atom in a separate molecule or within the same molecule.
- ==Hydrogen bonds are weaker than covalent bonds, but they are still strong enough to influence the behavior and properties of molecules.
They are responsible for many important biological processes, such as the binding of DNA strands in the double helix, the structure and function of proteins, and the properties of water==. - The strength of a hydrogen bond depends on several factors, including the distance between the two electronegative atoms, the angle between the hydrogen atom and the two electronegative atoms, and the strength of the electrostatic forces between the atoms.
In general, the closer and more aligned the two electronegative atoms are, the stronger the hydrogen bond. - Hydrogen bonds play a key role in determining the three-dimensional structure of biological molecules, such as proteins and nucleic acids.
They also contribute to the properties of water, such as its high boiling point, surface tension, and ability to dissolve many polar and charged substances.
IMPORTANTE
Slides with Notes