Questions
- What is the Proteolytic Cleavage?
- ==Proteolytic cleavage, also known as proteolysis, is a process by which proteins are broken down into smaller fragments or peptides through the action of proteases, enzymes that specifically cleave peptide bonds between amino acid residues==.
- ==Proteolytic cleavage is an important mechanism for regulating protein activity, as it can activate or inactivate proteins by removing or altering specific regions of the protein==.
In some cases, proteolytic cleavage can also lead to the release of functional domains or signaling peptides that can have biological activity on their own. - Proteolytic cleavage can occur in a variety of cellular compartments, including the cytoplasm, nucleus, endoplasmic reticulum, and extracellular space.
The specific proteases involved in proteolytic cleavage can vary depending on the protein and the cellular context, and may include enzymes such as caspases, calpains, and matrix metalloproteinases. - Proteolytic cleavage can have important roles in various biological processes, including development, cell signaling, and disease.
For example, proteolytic cleavage of the precursor protein amyloid precursor protein (APP) can generate amyloid beta peptides that are implicated in the pathology of Alzheimer’s disease.
—————————————————————
IMPORTANTE
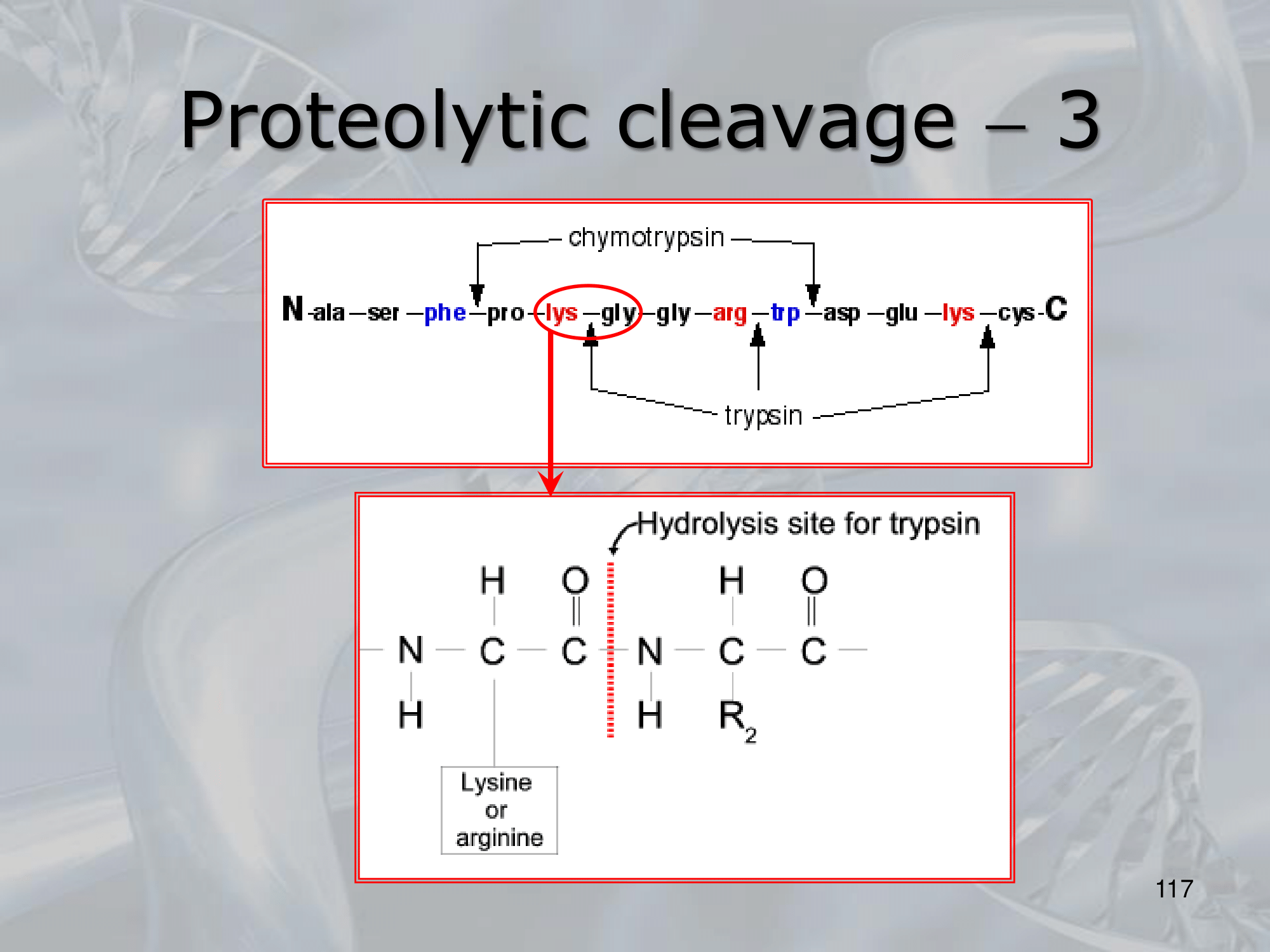
IMPORTANTE Proteolytic Cleavage: Both prokaryotes and eukaryotes posses several enzyme for cutting the proteins and peptides. There are different types of proteolytic cleavage:
- Removal of the methadine reside present at the start of each polypeptide (since the start codon also codes for methadine)
- Removal of the singal peptides
Sometimes the cleavage signal is constituted by a single residue. However, in many cases the sequence motif is longer and ambiguos, luckily with a NN we can get a prediction accuracy above .
Here are some example of ambiguos sequence motif
- Chymotrypsin cuts polypeptides at the C-terminal of bulky aromatic residues (~ex.: phenylalanine)
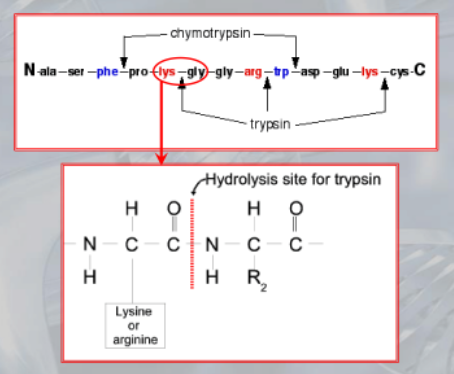
- Trypsin cuts the peptide bond on the carboxyl side of lysine and arginine residues
- Elastase cuts the peptide bond on the C-terminal of small residues (~ex.: glycine and alanine)
—————————————————————
Slides with Notes