Questions
- What is the Two-Dimensional Electrophoresis Technique?
- ==Two-dimensional electrophoresis (2-DE) is a widely used technique for separating complex protein mixtures based on their isoelectric point (pI) and molecular weight==.
The technique involves two separate electrophoretic separations, with the first dimension separating the proteins based on their pI, and the second dimension separating the proteins based on their molecular weight. - The first dimension separation is achieved using isoelectric focusing (IEF), a technique that separates proteins based on their net charge.
==The proteins are placed onto a strip of a gel matrix with a pH gradient, and an electric field is applied, causing the proteins to migrate towards the point on the gel with a pH equal to their pI.
As a result, the proteins are separated based on their pI==. - The second dimension separation is achieved using sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), a technique that separates proteins based on their molecular weight.
==The proteins are first denatured and then separated based on their size by running them through a polyacrylamide gel matrix in the presence of SDS, a detergent that binds to the proteins and imparts a negative charge.
As a result, the proteins are separated based on their molecular weight==. - After the two-dimensional separation is complete, the separated proteins can be visualized by staining the gel with a protein stain such as Coomassie Blue or by transferring the proteins onto a membrane for further analysis, such as Western blotting.
- Two-dimensional electrophoresis is a powerful technique for studying the proteome of a biological sample and has been used extensively in fields such as biomarker discovery, disease diagnosis, and drug discovery.
- ==Two-dimensional electrophoresis (2-DE) is a widely used technique for separating complex protein mixtures based on their isoelectric point (pI) and molecular weight==.
—————————————————————
IMPORTANTE
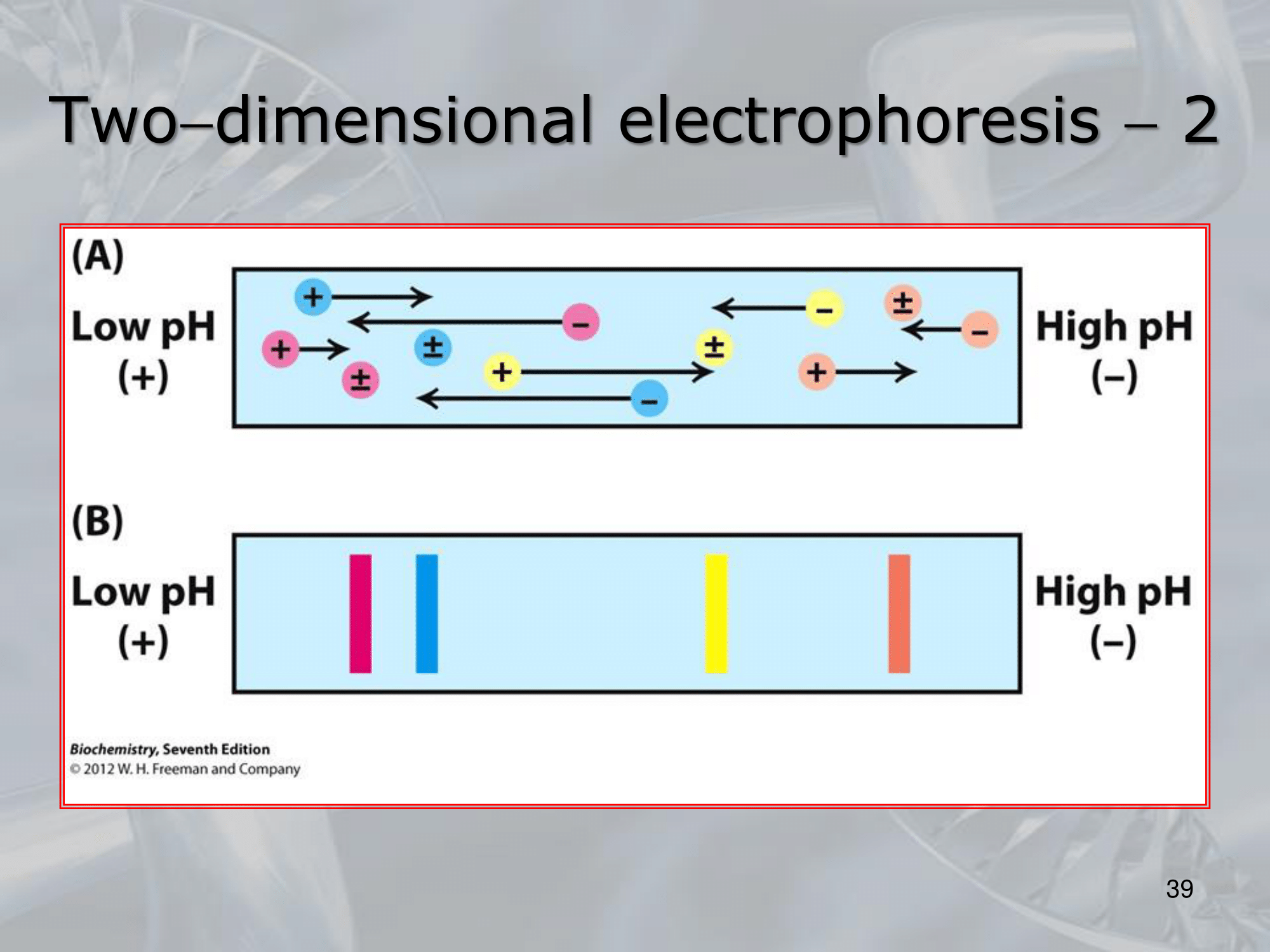
IMPORTANTE Two-Dimensional Electrophoresis: Allows the separation of proteins according to their molecular weight and charge. #IMPORTANTE The “first dimension is called” isoelectric focusing, the proteins collected from a tissue are placed on a polymeric support strip and are immersed in an immobilazing gradient of acidity, the protein will then move according to their pH to reach what is called an ==isoelectric point, which is a value of the pH at which a particular molecule carries no net electical charge==
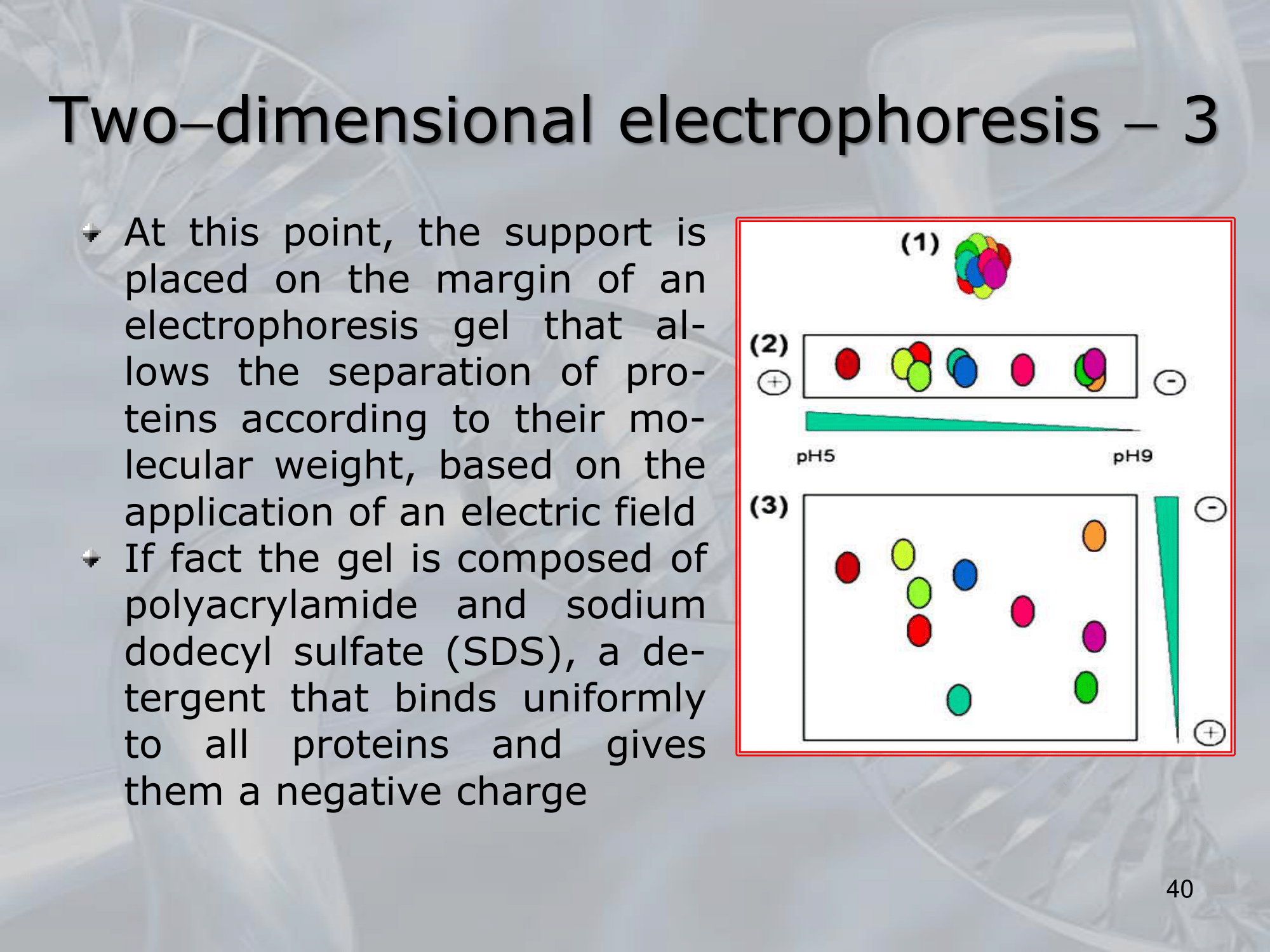
#IMPORTANTE At this point the support is placed on the margin of an electrophoresis gel, we “add the second dimension” which will focus the molecular weigth of the protein, an electric field is applied to the gel and the again the proteins will move reaching a point of stability:
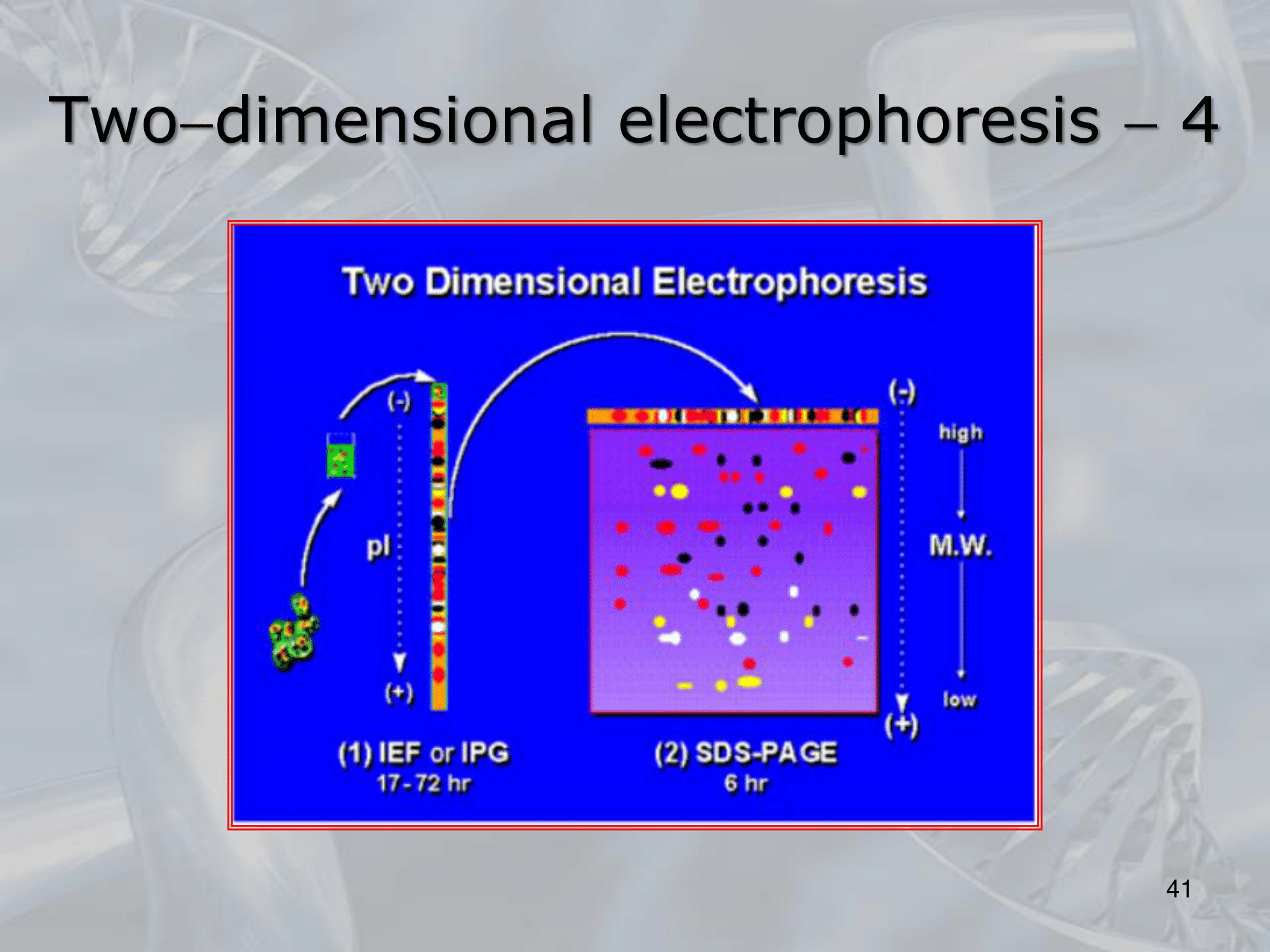
~Ex.: of an actual two-dimensional electrophoresis
#IMPORTANTE The last step is that of isolating the protein we are intrested in, either manually or we study it directly on the gel. Also we can study the resulting image of the elecrophoresis, seeing if a particular tissue either sane, diseased or treated (with respect to the same tissue is a different situation -sane, diseased or treated-) have an abbundace, lack or presence of some proteins. #IMPORTANTE There are some limitiation to this method. Mainly the diffuclty of identifing each protein at each dot (the human genome encodes many tens of thousands of proteins). Also the 2D electrophoresis is inadequate for the analysis of proteins that are very small or endowed with a little electric charge, but unluckly these proteins play an important role in many diseases. #IMPORTANTE It has some limitations but up to proteins can be found and analyzed in a singe tissue using this method.
—————————————————————
Slides with Notes

IMPORTANTE Two-Dimensional Electrophoresis: Allows the separation of proteins according to their molecular weight and charge. #IMPORTANTE The “first dimension is called” isoelectric focusing, the proteins collected from a tissue are placed on a polymeric support strip and are immersed in an immobilazing gradient of acidity, the protein will then move according to their pH to reach what is called an ==isoelectric point, which is a value of the pH at which a particular molecule carries no net electical charge==
#IMPORTANTE At this point the support is placed on the margin of an electrophoresis gel, we “add the second dimension” which will focus the molecular weigth of the protein, an electric field is applied to the gel and the again the proteins will move reaching a point of stability:
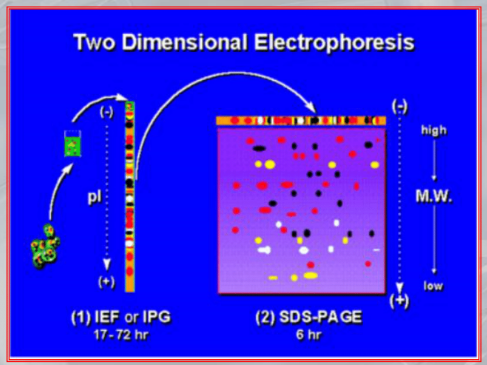
~Ex.: of an actual two-dimensional electrophoresis

#IMPORTANTE The last step is that of isolating the protein we are intrested in, either manually or we study it directly on the gel. Also we can study the resulting image of the elecrophoresis, seeing if a particular tissue either sane, diseased or treated (with respect to the same tissue is a different situation -sane, diseased or treated-) have an abbundace, lack or presence of some proteins. #IMPORTANTE There are some limitiation to this method. Mainly the diffuclty of identifing each protein at each dot (the human genome encodes many tens of thousands of proteins). Also the 2D electrophoresis is inadequate for the analysis of proteins that are very small or endowed with a little electric charge, but unluckly these proteins play an important role in many diseases. #IMPORTANTE It has some limitations but up to proteins can be found and analyzed in a singe tissue using this method.