Questions
- What are Inhibitors?
- Inhibitors are molecules that bind to a protein or enzyme and reduce or block its activity.
They can be natural or synthetic compounds that interfere with the normal function of the protein or enzyme. - There are several types of inhibitors, including:
- Reversible inhibitors: These inhibitors bind to the enzyme or protein in a non-covalent manner and can be displaced by other molecules.
Reversible inhibitors can be competitive, non-competitive, or uncompetitive, depending on their mode of action. - Irreversible inhibitors: These inhibitors bind to the enzyme or protein in a covalent manner, resulting in permanent inactivation.
Irreversible inhibitors are often used in the development of drugs for treating diseases. - Allosteric inhibitors: These inhibitors bind to a site on the protein or enzyme that is separate from the active site, causing a conformational change that reduces or blocks its activity.
- Reversible inhibitors: These inhibitors bind to the enzyme or protein in a non-covalent manner and can be displaced by other molecules.
- Inhibitors can be used for a variety of applications, including the development of drugs for treating diseases.
For example, inhibitors of enzymes involved in cancer cell growth can be used to treat cancer. Inhibitors can also be used as research tools for studying protein function and biological pathways.
- Inhibitors are molecules that bind to a protein or enzyme and reduce or block its activity.
- What is Drug Design?
- Drug design is the process of discovering and developing new drugs or improving existing drugs to treat various diseases and medical conditions.
It involves the use of computational and experimental methods to design molecules that can specifically target biological molecules, such as proteins or nucleic acids, involved in disease pathways. - Drug design typically involves the following steps:
- Identification of a target: This involves identifying a biological molecule that plays a key role in the disease pathway and is a potential target for drug therapy.
- Lead identification: This involves identifying or designing a molecule that can interact with the target and modify its activity. This molecule is called a “lead” compound.
- Lead optimization: This involves modifying the chemical structure of the lead compound to improve its properties, such as potency, selectivity, and pharmacokinetics.
- Preclinical testing: This involves evaluating the safety and efficacy of the optimized compounds in animal models.
- Clinical trials: This involves testing the safety and efficacy of the lead compounds in humans in a series of clinical trials.
- Regulatory approval: This involves obtaining regulatory approval from government agencies, such as the FDA, for the use of the drug in humans.
- Overall, drug design is a complex and iterative process that requires interdisciplinary collaboration between chemists, biologists, pharmacologists, and clinicians.
- Drug design is the process of discovering and developing new drugs or improving existing drugs to treat various diseases and medical conditions.
—————————————————————
IMPORTANTE
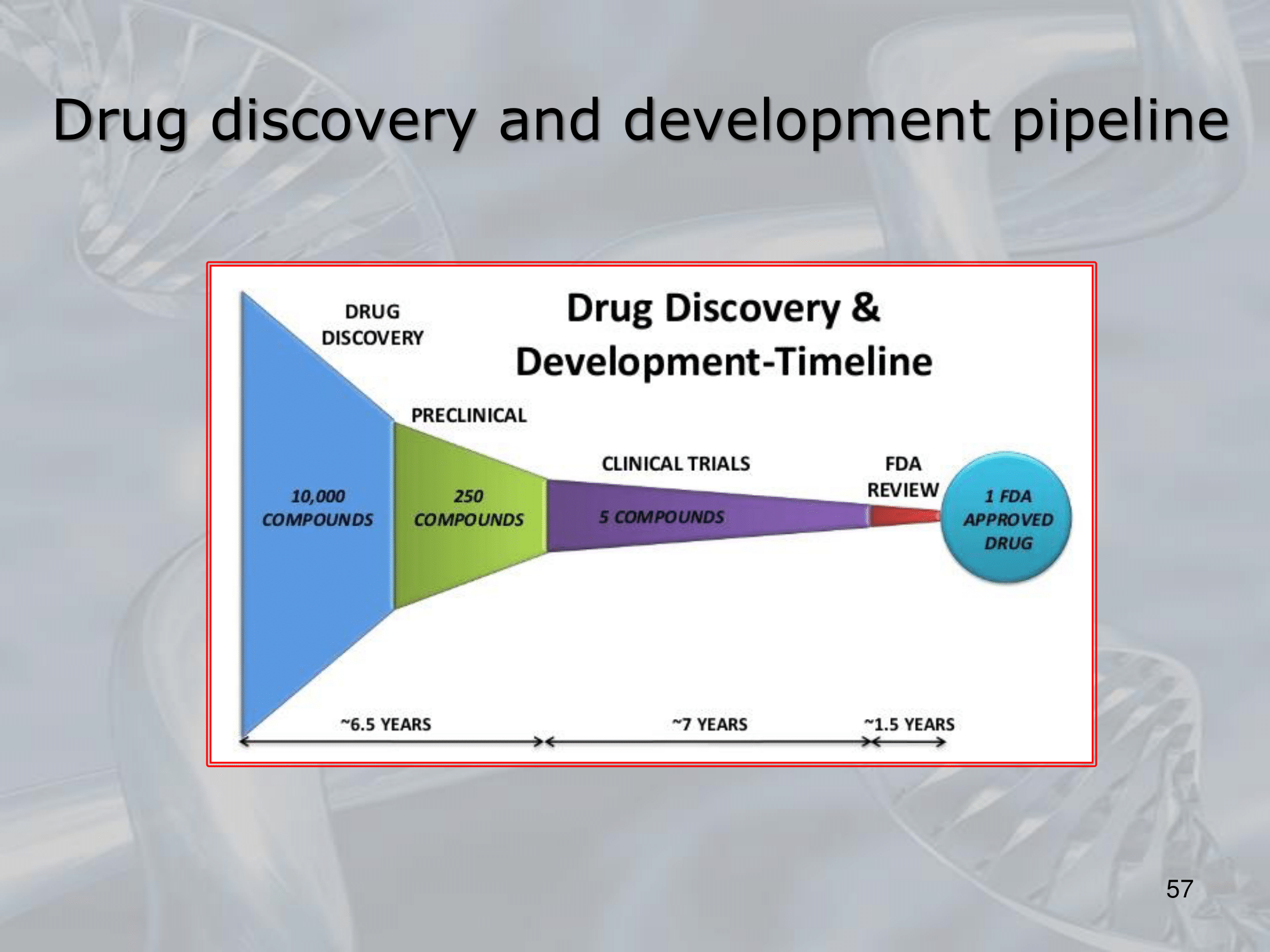
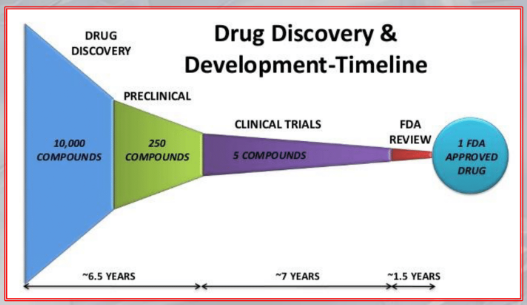
IMPORTANTE The development and testing of new drugs takes up to 15 years, and hundreds of million of euros.
IMPORTANTE Typically, researchers discover new drugs through:
- New insight into a disease process, allowing them to design a product to stop or reverse the effects.
- “Testing a lot of drugs on a lot of diseases”: many test of molecular compounds, to find possible beneficial effects against any of a large number of diseases
- Existing treatments that have unanticipated effects
- New technologies.
—————————————————————
Slides with Notes

IMPORTANTE The development and testing of new drugs takes up to 15 years, and hundreds of million of euros.
IMPORTANTE Typically, researchers discover new drugs through:
- New insight into a disease process, allowing them to design a product to stop or reverse the effects.
- “Testing a lot of drugs on a lot of diseases”: many test of molecular compounds, to find possible beneficial effects against any of a large number of diseases
- Existing treatments that have unanticipated effects
- New technologies.