Questions
- What is the Mass Spectromery Technique?
- Mass spectrometry (MS) is a powerful analytical technique used for identifying and quantifying molecules based on their mass-to-charge ratio.
In the context of proteomics, MS is used for the identification and quantification of proteins and their post-translational modifications. - The basic principle of MS involves ionizing a molecule and then separating and detecting the resulting ions based on their mass-to-charge ratio.
The ionization process can be achieved using a variety of techniques, including electrospray ionization (ESI), matrix-assisted laser desorption/ionization (MALDI), and others.
Once ionized, the resulting ions are separated based on their mass-to-charge ratio using a mass analyzer, such as a quadrupole, time-of-flight (TOF), or orbitrap.
The separated ions are then detected and measured, providing information on the mass and abundance of each ion. - In proteomics, MS is often coupled with liquid chromatography (LC), a technique for separating complex protein mixtures based on their physical and chemical properties.
This combination, known as LC-MS, allows for the identification and quantification of thousands of proteins in a single experiment.
In addition, MS can be used to identify post-translational modifications (PTMs) of proteins, such as phosphorylation, glycosylation, and acetylation, by detecting and characterizing the modified peptides. - MS has revolutionized the field of proteomics, enabling the identification and quantification of large numbers of proteins and PTMs from complex biological samples.
MS-based proteomics has a wide range of applications, including biomarker discovery, drug discovery, and understanding biological pathways and mechanisms.
- Mass spectrometry (MS) is a powerful analytical technique used for identifying and quantifying molecules based on their mass-to-charge ratio.
—————————————————————
IMPORTANTE
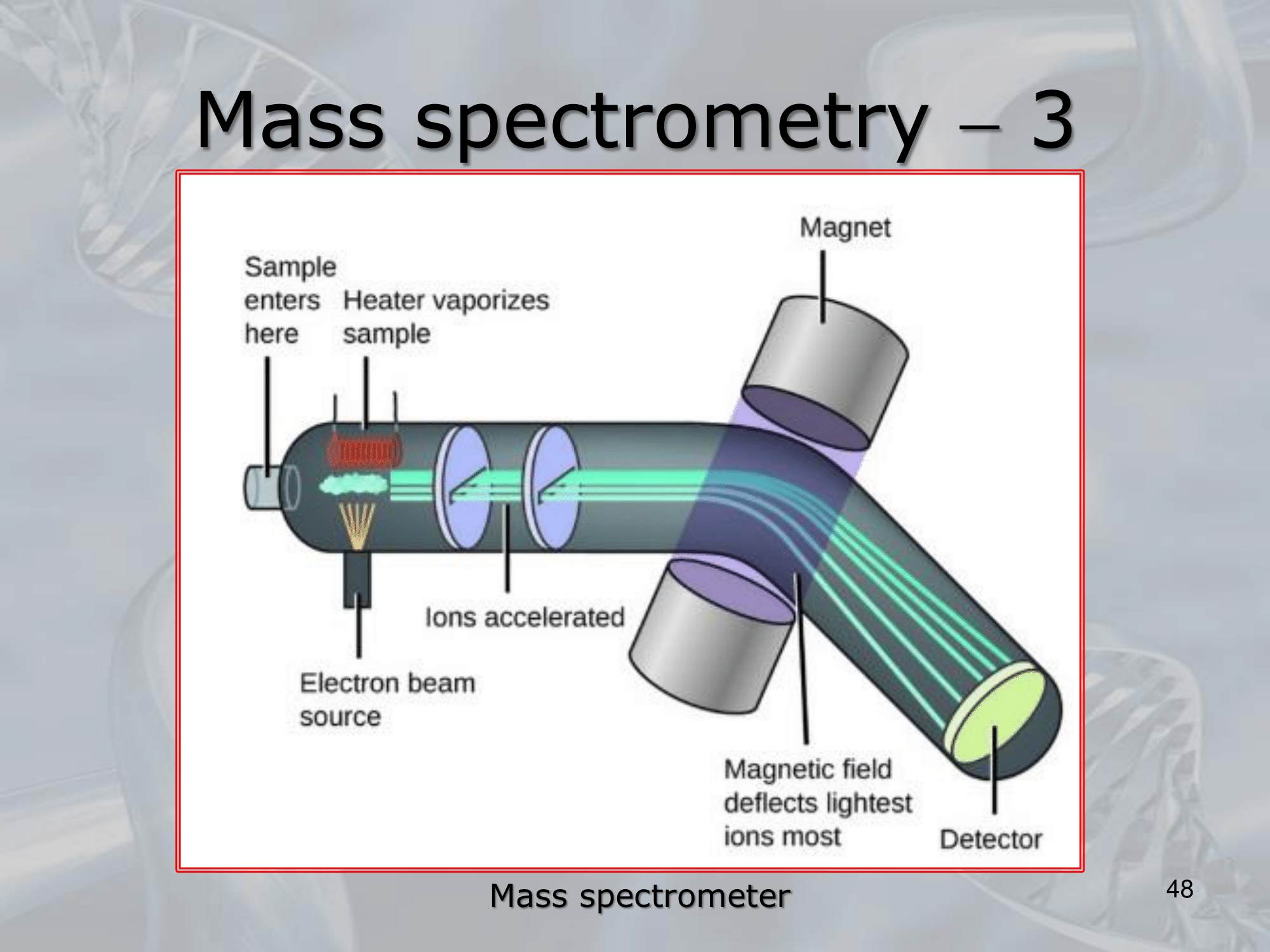
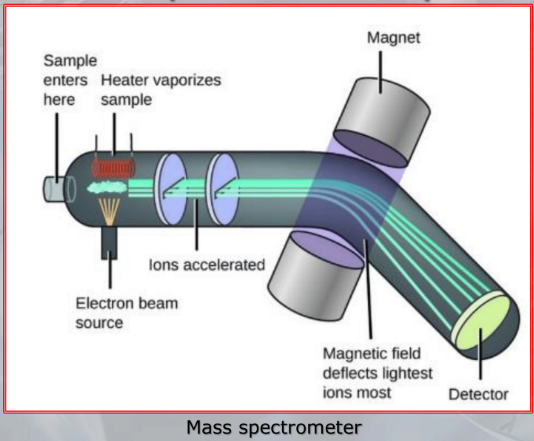
IMPORTANTE Mass Spectrometry: In mass spectometors samples may be introduced in any state (solid, liquid, gas) If the substance is solid or liquid it needs to be made volatile, before entering the ionization phase, during which the molecules of the compound are ionized, in the most common case by the interaction with an electron beam The ionized molucules are then “curved” by a magnetic field, finally these molecules collide with a “collector” placed at the end of the analyzer, such collision generate an electrical current that can be registered and measured, we will obtain a series of peaks of current.
In the mass spectrum graph the -axis shows the values of the ratio. The -axis is the relative abundance of the analyzed ions.
—————————————————————
Slides with Notes