Remeber:
- The Pauli Principle states that: ==two Identical particles can’t occupy the same energy level simultaneusly==.
- ==The density of atoms in a solid formed by pure Silicon is ==.
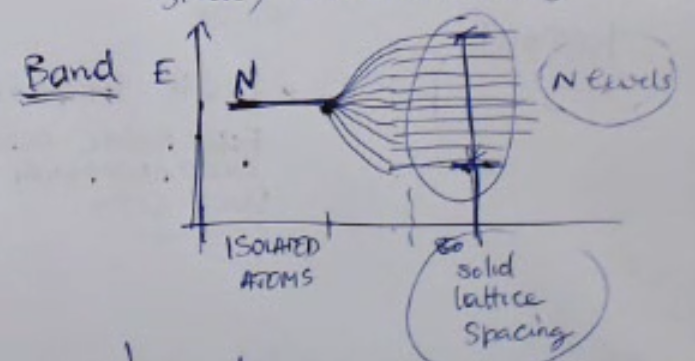
Pure Silicon means that there are no other atoms present in the solid.- Here is a graphical representation of the Pauli principle:
- The Silicon lattice is formed when the Silicon atoms are about distant from each other (Remember: ).
Memory Card

The Pauli Principle states that: ==two Identical particles can’t occupy the same energy level simultaneusly==.
Here we can see a graphical representation, where we take individual particles and “group” them toghter in a unic solid, while we move them closer togheter we can see the energy levels forming.
At the end when they are compleately groupped we will have levels, due to the pauli principle:
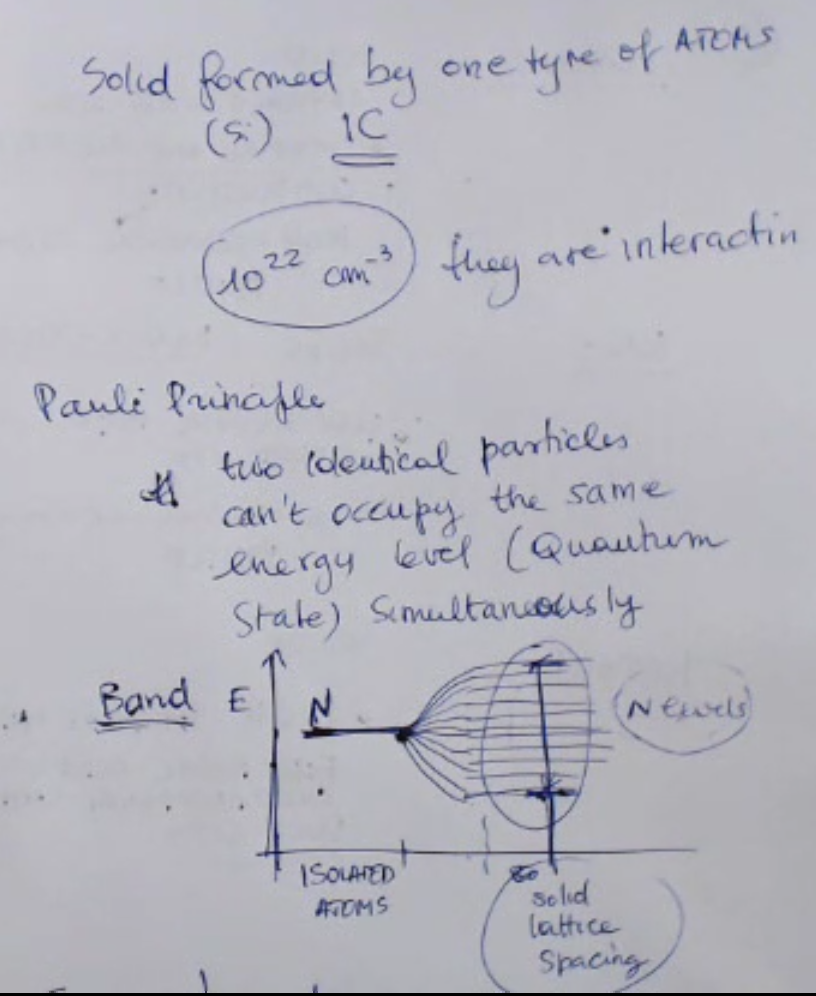
- Take into account that usually the density of atoms in a solid formed by pure Silicon (so no other atoms are present in this particular solid), is .
- ==So the Pauli principle tells us that two identical particles can’t occupy the same energy level or quantum state simultaneously==.
- If we form a solid, we put together and make a very large number of atoms interact with each other, the number of atoms which are bound together is so large that, due to the Pauli Principle, we will have so many different energy levels, (so due to the fact that they cannot be two identical levels in the same system), ==and these different energy levels are so close, dense and numerous that they can be considered as a continuum or a continuous interval of admitted levels, and these are called band==.