Remeber:
- Pure Silicon (Si) has electrons not belonging to a completed “shell”, so they can interact with each other.
Also to complete the outher shell a Silicon atom would need other electrons.- If we study how these electrons interact, if we change the distance between the atoms, at , we can see the forming of two “bands”:
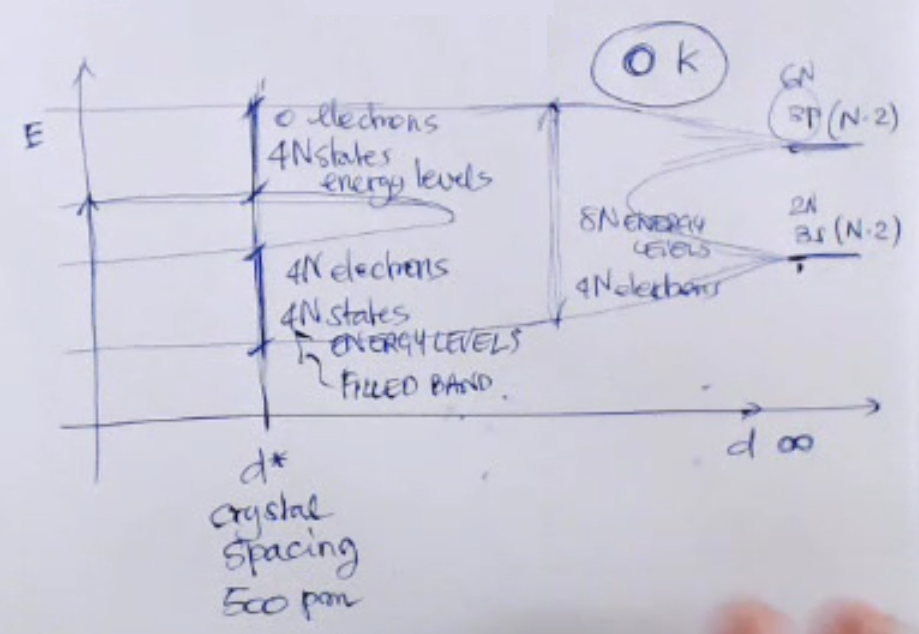
- Each band contains energy levels.
Where is number of particles in the system.
==If we were to take into consideration only atom (of Silicon), than ==.- Also the lower energy band is compleately filled, while the higher energy band is compleately empty.
- From this we can define:
- ==The valence band is the highest energy occupied band==.
- ==The conduction band is the lowest energy un-occupied band==.
- ==The band gap between valence band and conduction band where there are no allowed energy states==, for pure Silicon at this is about .
- Both the band gap and the probability of finding an electron in the conduction band change with temperature (see dependence of the fermi energy on temperature).
For example at the energy gap of Silicon is sligtly differnt: .
Memory Card

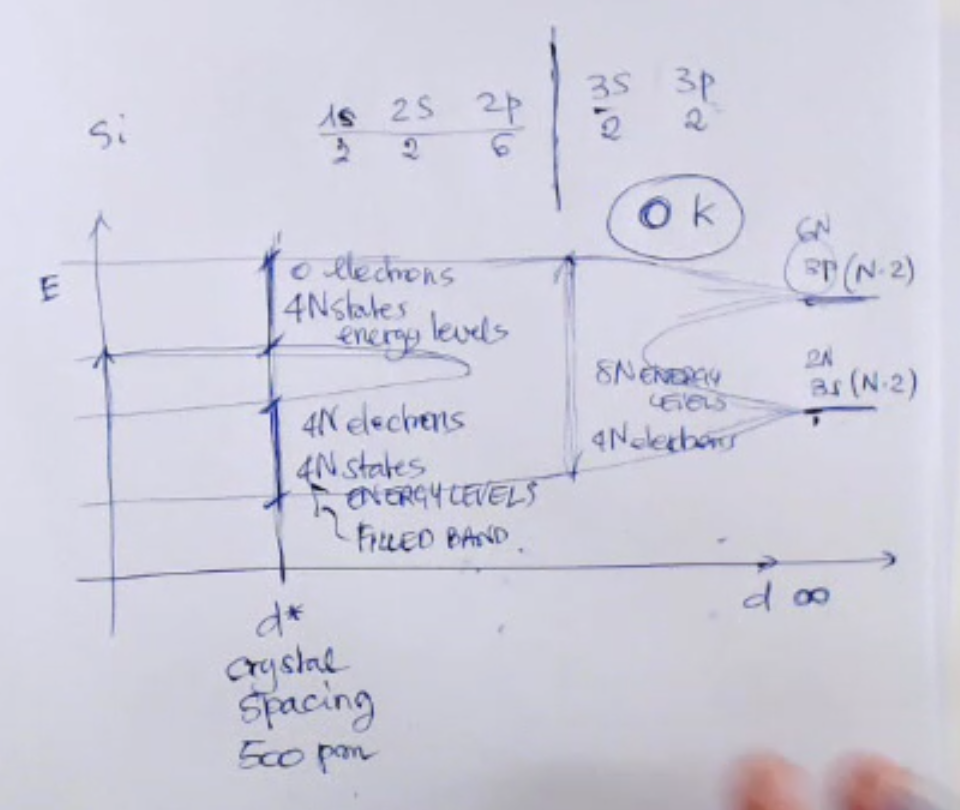
- So we have these (, , , , ) are the levels that form silicon.
- And the lowest energy level are: , ,
- So we have:
- : two electrons.
- : two electrons.
- : six electrons
- All of these are not interacting with other atoms.
- Instead the orbitals and the electrons which are involved in interaction are:
- : two electrons
- : two electrons.
- So the is a partially filled energy orbital, and these are the electrons responsible for interaction, and are the so called bond electrons.
- In the graph we have:
- : distance between atoms.
- : the right distance for the creation of the crystal, so for silicon this “crystal spacing” is about picometer.
- : energy.
- : number of particles in the system.
All orbital can contain up to electron.
All orbital can contain up to electron.
The same is true for the other orbitals in the whole system, or solid.
==If we take into consideration only atom (of Silicon), than ==.
- ==NOTE: we assume to be workiung at ==.
- So at infinite distance (), I have this energy for the level orbital and this energy for the orbital, we don’t care about the specifics.
Also I have both the and in the orbital contains electrons, where is completely filled, is partially empty. - When I start moving the atoms and bringing them closer, i see that energy bands start forming.
We end up with a total of energy lelvels.
So two bands (valence and conduction band) which span a total of energy levels.
But with only electrons (these are the bond electrons) that fills them. - At all bond electrons are found in the Valence Band.
If we were to increase the temperature than some electron would move from the Valence Band to the Conduction Band.
These are the definitions of the different bands and gap:

- ==The valence band is the highest energy occupied band==.
- ==The conduction band is the lowest energy un-occupied band==.
- ==The band gap between valence band and conduction band where there are no allowed energy states==.
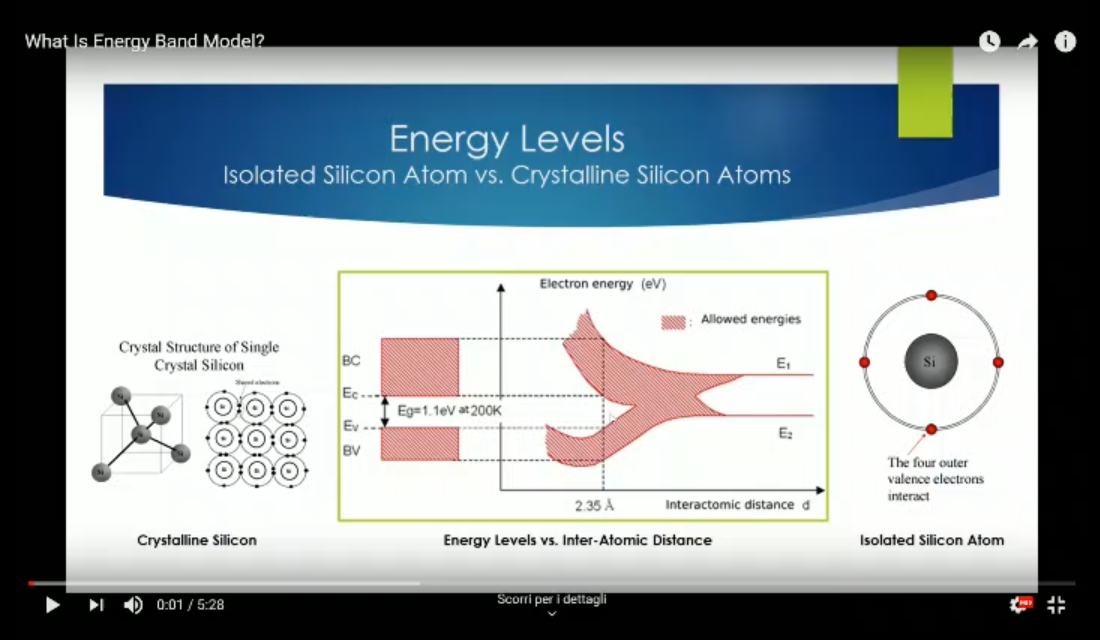
- Same as before, but a more accurate graph.